Phytochemical Composition and Biocontrol Potential of Chromolaena odorata Extracts against Brown Rot in Amber Sweet Orange Fruits During Ambient Storage
| Received 17 Nov, 2025 |
Accepted 30 Mar, 2026 |
Published 31 Mar, 2026 |
Background and Objective: Brown rot, a significant postharvest disease in tropical fruits, is increasingly attributed to the fungus Lasiodiplodia sp., which is notorious for causing fruit decay in citrus. Thus, the objective of this study was to assess the phytochemical compositions and biofungicide efficacy of Chromolaena odorata in controlling brown rot of orange fruits. Materials and Methods:Orange fruits infected with Lasiodiplodia were dipped into several concentrations of C. odorata extracts and disease severity was determined during ambient storage. Data collected on disease severity were subjected to Analysis of Variance and means of significant treatments were separated using Tukey’s Test at p<0.05. Results: Among the tested concentrations, only 10% of C. odorata extracts proved effective against the brown rot pathogen, with none of the fruit showing any sign of infection by day 30, while n-Hexadecanoic acid, Benzaldehyde 4-ethyl-, Hexanoic acid, 2, 3-Butanediol and Oleic acid occurred as the most abundant components in the extract. Conclusion: The findings of this study revealed that the aqueous extracts of C. odorata could be explored as natural, safe bio fungicide in citrus fruit preservation.
INTRODUCTION
Citrus fruits are hesperidium fruit type which belongs to the genus Citrusin the family Rutaceae. As a matter of fact, citrus production far exceeds that of any other fruit crop and it is widely grown in Nigeria and many other tropical and sub tropical regions1. Meanwhile, sweet orange is the most commercially planted citrus in Nigeria with a total production of 2.9 million tonnes in the year 20192. In fact, citrus especially sweet orange is a good source of vitamin C, folic acid and fibres. In fact, Turner and Burri3 reported that citrus fruits contain variety of vitamins, minerals, fibres anf phytochemicals such as carotenoids, flavonoids and limonoids which appera to have biological activities and health benefits. However, one of the greatest problems encountered during storage of sweet orange fruits is pathological deterioration. Thus, the potential foreign exchange earnings through the export of citrus fruits (sweet oranges) are under serious threat due to post-harvest infections. Regrettably, more than 50% of fruits grown in Nigeria are lost while moving from farms to major city markets4.
The two most important postharvest diseases in all citrus production are green rots caused by Penicillium digitatum and blue rots caused Penicillium italicum5. However, most losses associated with citrus production are caused by the green rot which accounts for up to 90% of the total citrus losses, especially in arid zones and subtropical climates6,7. However, Lasiodiplodia is a cosmopolitan fungus that is polyphagous and widespread, unspecialized virulent rot pathogen, found in the tropics and subtropics on a variety of host plants ranging from tree crops like kola, cocoa, to vegetables and fruits like orange, avocado, mango and cherry. In fact, Oladele and Aborisade8 reported Lasiodiplodia sp. IMI503248 as a major post-harvest pathogen of orange fruits in Akure, Nigeria, accounting for over 75% disease incidence of the total rots in their study. The fungus was identified by ITS rDNA sequence analysis using the FASTA algorithm with the fungus database from the European Molecular Biology Laboratory (EMBL) and the sequence showed 100% identity to numerous ITS sequences described from different Lasiodiplodia species or their Botryosphaeria teleomorphs. Best matches with the fungus included sequences of Lasiodiplodia species reported in the peer-reviewed literature9 and all the known species are associated with various symptoms such as dieback, root rot, fruit rot and leaf spots among many others10. Initial infection shows as light brown discolouration on any area of the fruit surface. As the decay develops, the lesion becomes more brown, firm and slippery11.
Meanwhile, postharvest diseases in fruits have been controlled mainly by synthetic fungicides either as dips, sprays, fumigants, treated wraps and box liners or in waxes and coatings. However, synthetic preservatives have raised concerns regarding their potential health risks and environmental impact, underscoring the need for innovative and eco-friendly solutions12. Besides, export markets are increasingly more sensitive to the use of chemicals for disease control coupled with the fact that most chemicals are expensive and inaccessible to local farmers who are the major bulk producers of this fruit in Nigeria. Development of alternative methods has become urgent in recent years due to the increasing regulatory restrictions on the use of fungicides13. Thus, promising alternatives include natural antimicrobials extracted from plants, animals or microorganisms that can suppress bacteria and fungi growth14.
Chromolaena odorata, commonly known as ‘ewe Awolowo’ and ‘obu inenawa’ by respective Yoruba and Ibo major tribes in Nigeria, has been reported in tradition medicine as antispasmodic, antiprotozoal, anti-trypanosomal, antibacterial, antifungal, antihypertensive, anti-inflammatory, astringent, diuretic and hepatotropic agent15. Despite its invasive nature, the plant has been found to have various traditional medicinal uses in different cultures, including wound healing, anti-inflammatory, and antimicrobial applications16.
Therefore the main objective of this study was to assess the effectiveness of C. odorata in controlling brown rot of orange fruits during ambient storage and its potential as a suitable alternative to synthetic preservatives.
MATERIALS AND METHODS
Source of fruits: Mature, green healthy orange fruits were harvested from a commercial orchard in September 2023 from a citrus farm in Igbatoro, Akure North, Nigeria. Fruits of uniform size and colour were selected. Before treatment, the fruits were washed with clean water, disinfected for 10 min in 10% sodium hypochlorite and allowed to air-dry at room temperature.
Preparation of spore suspension: A ten day old agar slant culture of Lasiodiplodia sp. (IMI Number: 503248) on malt extract agar (MEA) was used to prepare spore suspension. Sterile water was poured into the slant and shaken vigorously to dislodge the spores from the vegetative hyphae. The wash water was collected in a sterilized beaker. One milliliter of the suspension was spread on an area of 1 cm2 and allowed to dry on a clean microscope slide before counting spores using the formula of Breed Direct Counting Technique17 under the high dry ×40 objective microscope (Olympus).
Pathogenicity test: Spore suspension of Lasiodiplodia was used to inoculate fresh fruits 1 mm deep at the equator and incubated at 28±2ºC and 75% relative humidity inside sterilized desiccators. The disease symptoms were noted and re-isolation from infected fruit tissue was performed on fresh sterile MEA plate and its cultural characteristics were compared with the original isolate.
Preparation of extract of Chromolaena odorata: Detached Chromolaena odorataleaves were air dried for 14 days. The dried leaves were pulverized with a blender to a smooth powdery form. About 28 g of the pulverized leaves were dissolved in 1 L of water, thoroughly shaken together and later filtered with a muslin cloth to obtain the crude/stock solution. Varying concentrations (5, 10, 15 and 100%) of the aqueous extract were then prepared from the stock/crude extract with appropriate volumes of water.
Inoculation and treatment of orange fruits with Chromolaena odorata extract: The orange fruits were artificially infected with spores (6.50×104 CFU/mL) of Lasiodiplodia and later dipped separately into each concentration (5, 10, 15 and 100%) of the prepared extract for 5 min while benlated (fungicide) and untreated orange fruits served as positive and negative control, respectively. Each set up consisted of 3 fruits. After treatment, the fruits were then placed inside a sterilized Petri dish and transferred into desiccators, and stored at 28±2 and 75% relative humidity and assessed daily for disease severity.
Assessment of rot severity: Assessment of disease severity was done using the scale of Miller et al.18 but with slight modification and observed as follows:
| • | Disease free = 1 | |
| • | Slight rot/decay up to 10 % of the fruit = 2 | |
| • | Moderate rot/decay up to 25 % of the fruit = 3 | |
| • | Severe rots/decay ≥35 % of the fruit surface = 4 |
Rot/decay was recognized by light brown discolouration on the fruit or by appearance of mycelium on the fruit surface.
Determination of in vitro activity of the most effective concentration of Chromolaena odorata extract on spore germination of Lasiodiplodia sp.: Two millimeters of the spore suspension of the Lasiodiplodia sp. was collected into a beaker, followed by the addition of 2 mL of the most effective concentration (10%) of the Chromolaena odorata extract while spore suspension without extract (0%) served as control. After 12 hrs of incubation at 28±2°C, the mixed suspension was spread on an area of 1 cm2 and allowed to air-dry on a clean microscope slide containing solidified MEA in triplicates before viewing under ×10 objective binocular microscope (N-300M) to count the number of germinated and un-germinated spores.
Determination of in vitro activity of the most effective concentration of Chromolaena odorata extract on mycelia growth of Lasiodiplodia sp.: The effect of Chromolaena odorata extract on mycelia growth of Lasiodiolodia sp. was carried out using agar diffusion method. With 6mm cork-borer, two wells were bored at 2 cm apart on already solidified MEA. One well contained the most effective concentration (10%) of the extract while the other well contained 6 mm mycelia plug of the Lasiodiolodia sp. The set up was incubated at 28±2°C for 3 days. Each setup was in triplicates. After incubation, the mycelia growth was measured along the transect in two directions at right angle to each other.
Phytochemical screening of the aqueous extract of Chromolaena odorata: The phytochemical screening of the aqueous extract was done according to the method described by Harborne19. The phytochemicals screened for were tannin, saponin, phlobatinnin, flavoniod, alkaloid and cardiac glycosides.
|
Gas chromatography-mass spectrophotometry (GC-MS) of Chromolaena odorata extract: The Chromolaena extract was subjected to chromatographic analysis using a Varian 3800/4000 gas chromatograph mass spectrometer equipped with an Agilent Technologies and BP5 (30 m×0.25 mm×0.25 microns) capillary column. Nitrogen was used as a gas carrier. A 1.0 μL volumes of the extract were injected using a splitless mode at an injector temperature of 270°C. The oven temperature was ramped from 80 to 200°C (1 min hold) at a rate of 5°C/min. The oven temperature was held at 280°C for 6 min following each analysis. The total run time for each sample was approximately 45 min. The GC-MS interface temperature was set to 280°C. The peaks of the organic compounds in the samples were identified in Wiley’s NIST 08 Mass Spectral Library and expressed in terms of its balance and retention time.
Statistical analysis: The data obtained for the disease severity, spore germination and mycelia growth were subjected to analysis of variance and where significant, the means were compared at 5% level of probability using Tukey’s Test (SPSS version 20). The values are expressed as Mean±Standard Deviation.
RESULTS
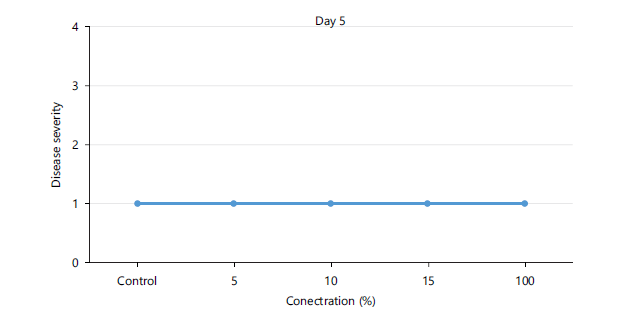
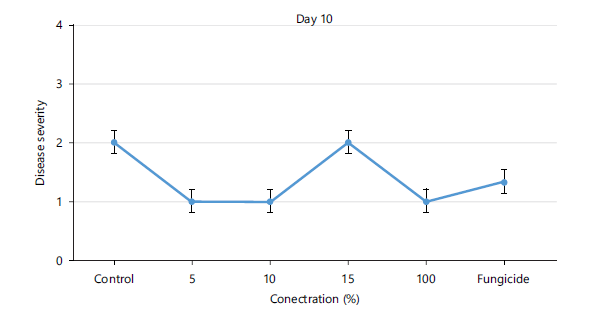
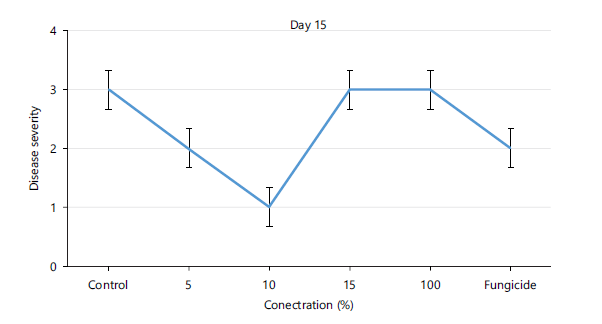
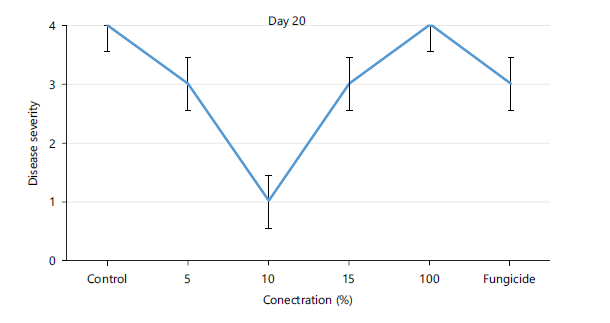
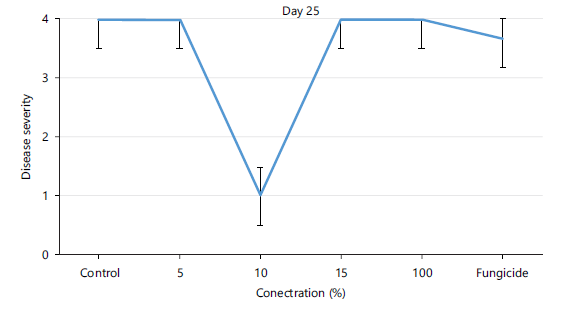
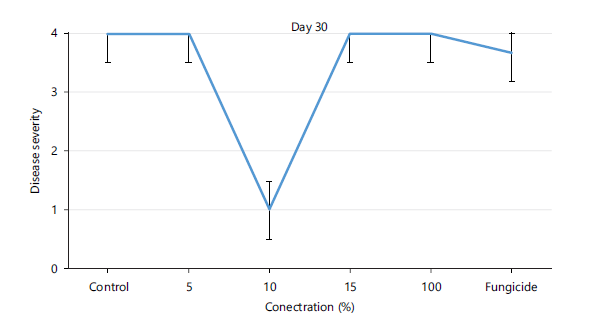
Disease severity of orange fruits infected with Lasiodiplodia sp. following treatment with different concentrations of Chromolaena odorata extracts: The observed results shown in Fig. 1 revealed that both the control (untreated) fruits and all the treated fruits with different concentrations (5, 10, 15, 100%) of the Chromolaena extract had the same disease severity (1.00±0.00) by day 5 of storage, indicating that they were all disease-free. As storage duration progressed till day 10, Fig. 2 showed that all treated fruits were still disease free, except the control and fruits treated with 15% extracts of C. odorata that had shown slight rottenness as evident by their mean disease severity values of 2.00±0.00. Even by days 15 and 20 in storage as observed in Fig. 3-4, respectively, fruits treated with 10%extractsstill maintained their diseased free status (1.00±0.00) when compared with fruits treated with 5% and 15% extract of C. odorata that showed moderate rottenness (3.00±0.03) while control and fruits treated with 100% C. odorata demonstrated complete rottenness (4.00±0.00). Likewise, as the storage durations became further extended to days 25 and 30, only fruits treated with 10% of C. odorata as shown in Plate 1 retained their non-diseased status (wholesomeness) of the orange fruits while Fig. 5-6 revealed that all other fruits (both control and treated fruits) had completely rotten.
|
|
|

|
|
|
|
| Table 1: | Effect of most effective concentrations of Chromolaena odorata extract on spore germination of brown rot pathogen in vitro | |||
| Most effective concentration of Chromolaen aodorata extract (%) |
Germinated spores (%) | Non-germinated spores (%) |
| 0 (control) | 115.67±12.81b | 13.33±9.61b |
| 10 | 10.33±5.55a | 71.67±2.33a |
| Means followed by same letters within columns are not significantly different (p>0.05) from one another using Tukey’s HSD Test | ||
| Table 2: | Effect of most effective concentrations of the aqueous extract of Chromolaena odorata on mycelia growth of the brown rot pathogen in vitro | |||
| Most effective concentrations of Chromolaena odorata extract (%) | Mycelia growth (cm) |
| 0 (control) | 1.20±0.12b |
| 10 | 0.83±0.12a |
| Means followed by same letters within the column are not significantly different (p<0.05) from one another using Tukey’s HSD Test | |
In vitro effect of Chromolaena odorata extract on spore germination and mycelia growth of the brown rot pathogen: The in vitro findings revealed that the percent spores that did not germinate following treatment with 10% extract of C. odorata was significantly higher (71.67±2.33) when compared with non-germinated spores (13.33±9.61) of the control (spores without extract treatment) (Table 1). Likewise, the brown rot pathogen had lesser mycelia growth (0.83±0.12 cm) when treated with 10% extract of C. odorata and was significantly different (p<0.05) from the untreated mycelia (Table 2).
Phytochemical compositions of Chromolaena odorata extracts: Tannins, alkaloids, flavonoids, saponins were all detected as phytochemical constituents in the extracts with the exception of cardiac glycosides that were absent (Table 3).
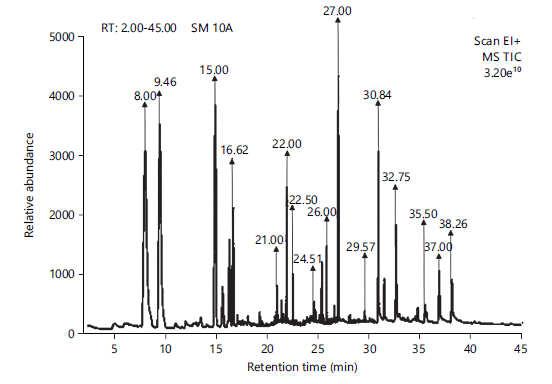
Bioactive compounds detected in Chromolaena odorata extracts: The chromatogram of aqueous extract of Chromolaena odorata using GC-MS is illustrated in Fig. 7. The peak area (%) of the various identified compounds included 2,3-Butanediol (10.09), Hexanoic acid (11.72), Benzaldehyde, 4-ethyl- (12.37), 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl- (7.16), Ethanone, 1-(2-hydroxy-5-methylphenyl)- (2.27), Methyl 4-(hydroxymethyl) Benzoate (7.82), p-Menth-3-en-9-ol (3.26), Eugenol (1.30), Acetamide, N-tetrahydrofurfuryl-2-methoxy- (4.56), n-Hexadecanoic acid (14.33), 9,12-Octadecadienoic acid methyl ester (1.04), Oleic acid (9.77), Octadecanoic acid (5.93), Squalene (1.37), Stigmasterol (3.58), and γ-Sitosterol (3.41) (Table 4). The analysis revealed the presence of n-Hexadecanoic acid (14.33%), Benzaldehyde 4-ethyl- (12.37%), Hexanoic acid (11.72%), 2, 3-Butanediol (10.09%) and Oleic acid (9.77%) as the major bioactive compounds in Chromolaena extract.
| Table 3: | Phytochemical constituents of aqueous extract of Chromolaena odorata | |||
| Phytochemicals | Chromolaena odorata extract |
| Tannins | + |
| Alkaloids | + |
| Cardiac glycosides | - |
| Saponins | + |
| Flavonoids | + |
| Table 4: | Bioactive compounds of Chromolaena odorata extract | |||
| S/N | Retention time (Min) | Compound detected | Peak area (%) |
| 1 | 8 | 2,3-Butanediol | 10.09 |
| 2 | 9.46 | Hexanoic acid | 11.72 |
| 3 | 15 | Benzaldehyde, 4-ethyl- | 12.37 |
| 4 | 16.62 | 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl- | 7.16 |
| 5 | 21 | Ethanone, 1-(2 hydroxy-5-methyl phenyl)- | 2.27 |
| 6 | 22 | Methyl 4-(hydroxymethyl) Benzoate | 7.82 |
| 7 | 22.5 | p-Menth-3-en-9-ol | 3.26 |
| 8 | 24.51 | Eugenol | 1.3 |
| 9 | 26 | Acetamide, N-tetrahydrofurfuryl-2-methoxy- | 4.56 |
| 10 | 27 | n-Hexadecanoic acid | 14.33 |
| 11 | 29.57 | 9,12-Octadecadienoic acid methyl ester | 1.04 |
| 12 | 30.84 | Oleic acid | 9.77 |
| 13 | 32.75 | Octadecanoic acid | 5.93 |
| 14 | 35.5 | Squalene | 1.37 |
| 15 | 37 | Stigmasterol | 3.58 |
| 16 | 38.26 | y-sitosterol | 3.41 |
DISCUSSION
This study has shown the efficacy of aqueous leaf extracts of Chromolaena odoratain controlling brown rot of orange fruits during ambient storage. Remarkably, only fruits treated with 10% extract were still disease free by day 30 under ambient storage. Thus, this showed that 10% concentration of the Chromolaena extract was able to compete favourably with the other concentrations by extending the shelf life of the orange fruits for 30 days at ambient storage as against the untreated that had already deteriorated by day 20 in storage. This is supported by the works of Abdullah et al.20 who conducted a study highlighting the inhibitory effects of Chromolaena odorata extract against pathogenic fungi responsible for postharvest decay in fruits and this underscores the plant’s ability to combat microbial growth, making it promising for postharvest disease control. Even, Efunwoye et al.21 in their own work reported that the treatment observed to be most effective in reducing the rates of disease incidences, extending the shelf-life and preserving marketability attributes in Padma tomato fruits was by submerging the fruits inside the botanical extract of Chromolaena odorata.
Even the in vitro study further supports the antifungal efficacy of the 10% extract of C. odorata in controlling brown rot on orange fruits for 30 days by inhibiting spore germination and significantly reducing the pathogen's mycelial weight. This confirms the effectiveness of plant extracts in managing post harvest pathogen. This observation is in agreement with the works of Oladele22 who reported though on garlic powder, that the different weights of garlic powder apart from the control (0 g garlic) significantly inhibited the mycelia growths of Aspergillus, Rhizopus, Lasiodiplodia and Mucor species which are the major post-harvest pathogens tested in his study.
In fact, the antimicrobial activity of Chromolaena odorata extract is attributed to bioactive compounds such as flavonoids and alkaloids18. This observation is equally in agreement with the reports of Chiejina and Onaebi23 that Chromolaena odorata extracts possess alkaloids, phenols, tannins, saponins and flavonoids with antimicrobial activities against Geotrichum candidum. These compounds have been shown to interfere with the growth and development of pathogenic fungi, making Chromolaena odorata an effective natural agent for disease management. Each of these phytochemical constituents as enunciated by other authors, is connected with significant antimicrobial properties. Some of these compounds are believed to contribute to its medicinal properties, including its antifungal activity24.
For instance, alkaloids and flavonoids when presentexhibit antimicrobial properties. These compounds inhibit the growth of spoilage-causing microorganisms, extending the shelf life of fruits25. These compounds possess antioxidant properties, which play a crucial role in fruit preservation by inhibiting oxidative processes and prevent the degradation of fruits by scavenging free radicals that contribute to spoilage26. This is further buttressed by the reports of Cerioni and Rapisarda27 that alkaloids and flavonoids act as natural antimicrobial agents, inhibiting the growth of bacteria and fungi that contribute to fruit spoilage. Also, tannins act as natural preservatives by forming complexes with proteins and enzymes, thereby inhibiting microbial growth and enzymatic browning28. Recent research has focused on the plant's potential as a source of bioactive compounds, with studies identifying several flavonoids and terpenoids that may have pharmaceutical significance29.
A lot of bioactive compounds and these natural compounds exert their effects through various mechanisms, making them effective against different types of microorganisms. Also, 9-octadecenoic acid methyl ester according to Donia and Hamann30 possesses anti-microbial properties.
CONCLUSION
This study demonstrates the efficacy of Chromolaena odorata extracts in controlling brown rot in orange fruit and providing significant insights into alternative and sustainable methods for managing this fungal disease. From this study, the use of aqueous extract (10%) of Chromolaena odorata was able to prolong the shelf life of orange fruits at ambient storage at least for 30 days. The extracts exhibited promising antifungal activity and effectively curtailed the spread of brown rot, with a reduction in disease severity observed in treated fruits. The ability of C. odorata in inhibiting the growth of microorganism can be traced to the phytochemicals present in the extracts that have also been identified to exhibit bioactive properties. These extracts are biodegradable and present minimal risk to human health and the environment, positioning them as safer bio fungicide alternative to synthetic fungicides.
SIGNIFICANCE STATEMENT
This study explored the use of Chromolaena odorata extracts in managing brown rot disease in orange fruits. The results showed that the aqueous extracts of C. odorata significantly extended the shelf life of orange fruits, with 10% aqueous extracts outperforming other concentrations and the test fungicide by keeping the orange fruits healthy for at least 30 storage days. Thus, this research revealed eco-friendly, cheap, local and non-chemical approach to solving post management of plant diseases.
ACKNOWLEDGMENT
The authors thank Life Technologies, UK for PCR purification and sequencing reactions of the fungi isolate. Authors also acknowledge Federal University of Technology, Akure, Nigeria for providing necessary research facilities.
REFERENCES
- Piccinelli, A.L., M.G. Mesa, D.M. Armenteros, M.A. Alfonso, A.C. Arevalo, L. Campone and L. Rastrelli, 2008. HPLC-PDA-MS and NMR characterization of C-glycosyl flavones in a hydroalcoholic extract of Citrus aurantifolia leaves with antiplatelet activity. J. Agric. Food Chem., 56: 1574-1581.
- Doki, N.O., C.I. Eya, M.F. Tughgba, O.G. Akahi and A. Ameh, 2019. Determinants of post-harvest losses of orange in selected local government areas of Benue State. Int. J. New Econ. Social Sci., 10: 295-308.
- Turner, T. and B.J. Burri, 2013. Potential nutritional benefits of current citrus consumption. Agriculture, 3: 170-187.
- Olife, I.C., O.A. Ibeagha and A.P. Onwualu, 2015. Citrus fruits value chain development in Nigeria. J. Biol. Agric. Healthcare, 5: 36-47.
- Wuryatmo, E., A. Klieber and E.S. Scott, 2003. Inhibition of citrus postharvest pathogens by vapor of citral and related compounds in culture. J. Agric. Food Chem., 51: 2637-2640.
- Macarisin, D., L. Cohen, A. Eick, G. Rafael, E. Belausov, M. Wisniewski and S. Droby, 2007. Penicillium digitatum suppresses production of hydrogen peroxide in host tissue during infection of citrus fruit. Phytopathology, 97: 1491-1500.
- Marcet-Houben, M., A.R. Ballester, B. de la Fuente, E. Harries, J.F. Marcos, L. González-Candelas and T. Gabaldón, 2012. Genome sequence of the necrotrophic fungus Penicillium digitatum, the main postharvest pathogen of citrus. BMC Genomics, 13.
- Oladele, O.O. and A.T. Aborisade, 2015. Integrated control of Lasiodiplodia rot on sweet orange fruits using hot water-fungicide treatment. Niger. J. Mycol., 7: 93-99.
- Rodriguez, R.J., J.F. White Jr., A.E. Arnold and R.S. Redman, 2009. Fungal endophytes: Diversity and functional roles. New Phytol., 182: 314-330.
- Slippers, B. and M.J. Wingfield, 2007. Botryosphaeriaceae as endophytes and latent pathogens of woody plants: Diversity, ecology and impact. Fungal Biol. Rev., 21: 90-106.
- Ismail, M. and J. Zhang, 2004. Post-harvest citrus diseases and their control. Outlooks Pest Manage., 15: 29-35.
- Christudas, S., T.M. Kulathivel and P. Agastian, 2012. Phytochemical and antibacterial studies of leaves of Tridax procumbens L. Asian Pac. J. Trop. Biomed., 2: S159-S161.
- Droby, S., M. Wisniewski, D. Macarisin and C. Wilson, 2009. Twenty years of postharvest biocontrol research: Is it time for a new paradigm? Postharvest Biol. Tech., 52: 137-145.
- Arshad, M.S. and S.A. Batool, 2017. Natural Antimicrobials, their Sources and Food Safety. In: Food Additives, Karunaratne, D.N. and G. Pamunuwa (Eds.), IntechOpen, London, UK, ISBN: 978-953-51-4662-9.
- Akinmoladun, A.C., E.O. Ibukun and I.A. Dan-Ologe, 2007. Phytochemical constituents and antioxidant properties of extracts from the leaves of Chromolaena odorata. Sci. Res. Essays, 2: 191-194.
- Omokhua, A.G., L.J. McGaw, J.F. Finnie and J. van Staden, 2016. Chromolaena odorata (L.) R.M. King & H. Rob. (Asteraceae) in Sub-Saharan Africa: A synthesis and review of its medicinal potential. J. Ethnopharmacol., 183: 112-122.
- Gross, T., J. Faull, S. Ketteridge and D. Springham, 1995. Introductory Microbiology. 1st Edn., Springer, New York, USA, ISBN: 978-1-4899-7194-4, Pages: 414.
- Miller, W.R., R.E. McDonald and J.L. Sharp, 1991. Quality changes during storage and ripening of 'Tommy Atkins' mangos treated with heated forced air. HortScience, 26: 395-397.
- Harborne, J.B., 1973. Phytochemical Methods: A Guide to Modern Techniques of Plant Analysis. 1st Edn., Chapman & Hall, Boundary Row, London, ISBN: 9780412105401, Pages: 278.
- Yaouba, A., E.K. Nnomo, A.S.Z. Medjap and L.C.E. Etoa, 2017. Antibacterial effect of Chromolaena odorata extracts on some Erwinia isolates, causal agents of tomato fruit rot in Dschang, Cameroon. Agric. Food Secur., 6.
- Efunwoye, O.O., A.T. Aborisade and O.O. Oladele, 2024. Treatment of tomato (Solanum lycopersicum L.) fruits with powders and ashes from Tridax procumbens and Chromolaena odorata for shelf-life extension. Afr. J. Agric. Food Sci., 7: 273-287.
- Oladele, O.O., 2019. Screening for antifungal activity of garlic (Allium sativum) powder against mycelia growth of three post-harvest pathogens. Eur. J. Biol. Res., 9: 57-63.
- Chiejina, N.V. and C.N. Onaebi, 2016. Phytochemical constituents and antifungal properties of Chromolaena odorata L. and Moringa oleifera Lam on fungal rot of cucumber (Cucumis sativus L.) fruit. Asian J. Plant Sci., 15: 35-41.
- Ingole, V.V. and S.R. Katad, 2024. Chemical composition, antioxidant, antibacterial activity of isolated oil and methanol extract of Tridax procumbens L. Int. J. Pharm. Sci. Res., 15: 1157-1166.
- Das, K., 2014. Phytochemical evaluation and comparative antibiocide efficacy of aqueous, ethanolic and equal mixture of aqueous and ethanolic (1:1) bark extract of Lannea coromandelica L. procured from Eastern Region of India. Int. Lett. Nat. Sci., 26: 21-31.
- Andriana, Y., T.D. Xuan, T.N. Quy, T.N. Minh, T.M. Van and T.D. Viet, 2019. Antihyperuricemia, antioxidant and antibacterial activities of Tridax procumbens L. Foods, 8.
- Cerioni, L. and V.A. Rapisarda, 2026. Management of postharvest fungal diseases in fruits and vegetables. J. Fungi, 12.
- Doughari, J.H., P.A. Ndakidemi, I.S. Human and S. Benade, 2012. Antioxidant, antimicrobial and antiverotoxic potentials of extracts of Curtisia dentata. J. Ethnopharmacol., 141: 1041-1050.
- Schmourlo, G., R.R. Mendonca-Filho, C.S. Alviano and S.S. Costa, 2005. Screening of antifungal agents using ethanol precipitation and bioautography of medicinal and food plants. J. Ethnopharmacol., 96: 563-568.
- Donia, M. and M.T. Hamann, 2003. Marine natural products and their potential applications as anti-infective agents. World Appl. Sci. J., 3: 338-348.
How to Cite this paper?
APA-7 Style
Oladele,
O.O., Oyedokun,
F.D., Fatukasi,
I.O. (2026). Phytochemical Composition and Biocontrol Potential of Chromolaena odorata Extracts against Brown Rot in Amber Sweet Orange Fruits During Ambient Storage. Asian Journal of Plant Pathology, 20(1), 1-11. https://doi.org/10.3923/ajpp.2026.1.11
ACS Style
Oladele,
O.O.; Oyedokun,
F.D.; Fatukasi,
I.O. Phytochemical Composition and Biocontrol Potential of Chromolaena odorata Extracts against Brown Rot in Amber Sweet Orange Fruits During Ambient Storage. Asian J. Plant Pathol. 2026, 20, 1-11. https://doi.org/10.3923/ajpp.2026.1.11
AMA Style
Oladele
OO, Oyedokun
FD, Fatukasi
IO. Phytochemical Composition and Biocontrol Potential of Chromolaena odorata Extracts against Brown Rot in Amber Sweet Orange Fruits During Ambient Storage. Asian Journal of Plant Pathology. 2026; 20(1): 1-11. https://doi.org/10.3923/ajpp.2026.1.11
Chicago/Turabian Style
Oladele, Oluwole, Olakunle, Feranmi David Oyedokun, and Ilesanmi Olajide Fatukasi.
2026. "Phytochemical Composition and Biocontrol Potential of Chromolaena odorata Extracts against Brown Rot in Amber Sweet Orange Fruits During Ambient Storage" Asian Journal of Plant Pathology 20, no. 1: 1-11. https://doi.org/10.3923/ajpp.2026.1.11

This work is licensed under a Creative Commons Attribution 4.0 International License.



