Epidemiology of the Major Fungal Diseases of Cashew Tree (Anacardium occidentale L.) in Togo
| Received 28 May, 2024 |
Accepted 04 Jul, 2024 |
Published 05 Jul, 2024 |
Background and Objective: Cashew tree cultivation is currently booming in Togo and their nuts are Togo’s fourth most important agricultural export. A study was initiated to draw up an inventory of cashew fungal diseases and determine their epidemiological parameters. Materials and Methods: Thirty two cashew orchards were surveyed, with eight orchards per prefecture. Twenty trees per orchard were randomly selected along the diagonals and inspected. Incidence was defined as the proportion of cashew trees showing symptoms of a fungal disease. Severity was assessed on an arbitrary five-grade scale by assigning quality values to infected organs. The severity index was determined by a mathematical formula. Prevalence was the proportion of localities affected by a fungal disease per prefecture. Results: Eight fungal diseases were inventoried, including anthracnose, black rust, bud dieback, yellow leaf spot, pestalotiosis, powdery mildew, gummosis and ramularia. Incidence and severity index were 61.09 and 36.56%, respectively for pestalotiosis, 38.12 and 20.16% for leaf anthracnose, 35.62 and 14.84% for yellow leaf spot and 30.47 and 20.46% for bud dieback. The prevalence of anthracnose leaf spot was the most widespread (96.88%), followed by yellow leaf spot (84.38%), pestalotiosis (84.38%) and black rust (81.25%). Conclusion: Because of the impact of these fungal diseases, it would be necessary to use phytosanitary products, improve agricultural practices and introduce more resistant cashew cultivars for new cashew orchards.
INTRODUCTION
Anacardium occidentale L. is a tropical fruit plant grown mainly for its nuts and apples. It is susceptible to pests and diseases, with symptoms visible on leaves, fruits, inflorescences, twigs, stems and roots1,2. Fungal diseases are the most common and diverse of cashew tree diseases. Several sanitary surveys conducted in cashew orchards have reported the presence of anthracnose, black rust, yellow leaf spot, bud dieback, powdery mildew, gummosis, pestalotiosis, red rust and bacteriosis1,3–5. The disruption to the harmonious development of cashew trees and the reduction in productivity are the direct impacts of these diseases. The latter is influenced by environmental conditions, cultivation practices, cashew varieties and pathogens virulence1. Anthracnose is most severe during the rainy season when temperatures range from 22 to 28°C and there are at least 10 hrs of saturated humidity6. According to Adejumo7, diseases and pests are one of the main reasons for low cashew yields. Cashew nut losses are estimated at 20-30% in Nigeria. Production losses due to anthracnose can be as high as 72%8, 50-70% for powdery mildew9,10and 48.4% for black rust11. Fusarium oxysporum, responsible for cashew fusariosis, can cause yield losses of up to 100% if left uncontrolled12. The damage to cashew fruits can reduce their quality and therefore their commercial value. This represents a loss of income for producers, particularly smallholders. Resistant cashew cultivars have been used to control anthracnose in Mozambique13. In Brazil, the selection of wild cashew genotypes resistant to gummosis, such as the CAPC 42 clone, has been the preferred means of controlling the pathogen14,15. The objectives of this study are to draw up an inventory of cashew fungal diseases in the prefectures (Anie, Est-mono, Tchamba and Tchaoudjo) of high cashew nuts production in Togo and to determine their incidence, severity, severity index and prevalence.
MATERIALS AND METHODS
Study area: Phytosanitary surveys were carried out in cashew orchards in the prefectures of Anie, Est-Mono, Tchamba and Tchaoudjo. The area lies between Latitudes 7°10 and 9°20 North and Longitudes 0°90 and 1°40 East. Throughout the year, the average temperature in the prefectures of Est-Mono and Anie is 26.1°C, with an average rainfall of 1001.6 mm. Relative humidity is around 43% in the hottest month (January) and 86% in the coldest (July or August). Depending on the month, the wind speeds vary from 3.1 to 4.4 km/hr. In the prefectures of Tchamba and Tchaoudjo, the average temperature is 26.4°C and the average annual rainfall is 912.3 mm. Relative humidity ranges from 32% in the warmest month (January) to 86% in the coldest month (August). Wind speeds annually vary from 2 to 2.8 km/hr. Four different soil types were reported in this study area. Tropical ferruginous soils (leached with concretions and curasse), Vertisols and Paravertisols were found in all four prefectures. Poorly eroded soils were only found in the Tchaoudjo prefecture. The prefectures are all located in Ecological Zone III, with the exception of the Tchaoudjo prefecture, which straddles ecological zones II and III. Ecological zone II (northern mountains) has a vegetation composed of savannahs and open and dry forests while ecological zone III (Central Plains) includes Guinean wooded savannahs and open and dry forests.
Fungal diseases inventory: Cashew-growing areas and lists of producers were documented with the support of Togolese Institute of Agronomic Research (ITRA). A total of 32 peasant cashew orchards of at least 2 ha were randomly selected and surveyed. These orchards were proportionally distributed among the four prefectures. The phytosanitary surveys were conducted in February, 2022, during the cashew fruiting phase. Twenty cashew trees at least 10 m apart were randomly selected along the diagonals of each peasant cashew orchard and inspected. The fungal diseases inventory was based on a search for fungal symptoms previously reported on cashew trees2-4.
Epidemiological parameters determination
Incidence: Incidence was the proportion of cashew trees showing the symptom of a target fungal disease. It was determined using the following formula1:
Where:
| I | = | Incidence | |
| As | = | Number of cashew trees symptomatic with the target fungal disease | |
| Api | = | Total number of inspected cashew trees. |
Severity: Severity was assessed on an arbitrary grade of five degrees by assigning quality grades to infected organs (Table 1) according to the modified method of Domedjui et al.1.
| Table 1: | Grading scale for fungal diseases of cashew | |||
| Grades | Infected surface (%) | Symptom’s interpretation |
| 0 | 0 | Absent or healthy plant |
| 1 | 01-10 | Minor |
| 2 | 10-30 | Slight |
| 3 | 31-60 | Moderate |
| 4 | 61-90 | Severe |
| 5 | 91-100 | Very severe or general necrosis |
Severity index: The severity index was determined using the adapted Francis et al.16 formula:
Where:
| Is | = | Fungal disease severity index | |
| Xi | = | Severity i of fungal disease on a cashew tree | |
| ni | = | Number of cashew trees with a severity I | |
| N | = | Total number of inspected cashew trees | |
| Z | = | Highest severity scale, i.e., 5 |
Prevalence: The proportion of localities affected by a fungal disease in an area. It was determined using the following formula1:
Where:
| P | = | Prevalence | |
| Lt | = | Number of localities affected by a fungal disease | |
| Lp | = | Total number of localities surveyed |
Data processing and statistical analysis: The geographical coordinates of the surveyed orchards were entered into Excel 2016 and the survey area was mapped using QGIS 2.18.14 software. The fungal disease pictures were processed using Paint 3D 6.2305.16087.0 software. Incidence, severity, severity index and prevalence data were imported into Excel 2016 and R software version 4.0.5 was used for statistical analysis. Analysis of Variance (ANOVA) and comparisons of means were performed using Tukey's test at the 5% significance level.
RESULTS
Phytosanitary surveys: Cashew cultivars with red, light red, yellow and golden yellow apples were grown and were at the fruiting stage during the survey. In several cashew orchards, mixtures of cashew cultivars have been observed. The orchards’s age varied from five to forty-eight years. The spacing commonly observed in the orchards varied between 10×10 and 14×14 m. These spacings allowed for intercropping of crops such as soybean, maize, sesame, groundnut, cassava, sorghum, cowpea, tomato, vegetable cornet, cabbage, okra and yam. Most of the orchards surveyed were located away from watercourses. Sanitary measures applied in the event of infection were limited to pruning infected branches. Plant debris resulting from the pruning of infected branches or crops is burnt on site or buried in the ground. Most of the orchards inspected were part of cashew orchards or surrounded by crops.
Fungal diseases inventoried: A total of eight fungal diseases were inventoried during the phytosanitary surveys. These were anthracnose (leaves and fruits), black rust, bud dieback, yellow leaf spot, pestalotiosis, powdery mildew, gummosis and ramularia.

|

|

|
On the leaves, anthracnose was characterized by the presence of irregular orange-brown spots visible on both sides (Fig. 1a). Infected fruits showed dark brown rot, while inflorescences were dry (Fig. 1b).
Necrosis of young shoots was characteristic of bud dieback (Fig. 2a). These young shoots were then covered with a powdery white felting (Fig. 2b).
Gummosis was characterized by one or more discharges of thick amber exudates on the trunks (Fig. 3a) or branches of cashew tree (Fig. 3b).
Necrosis of immature fruits was characteristic symptom of black rust (Fig. 4a-b).
Yellow leaf spot was characterized by the presence of numerous pale-yellow spots with irregular margins, varying in size and visible on both sides of the leaves (Fig. 5a-b).
Powdery mildew infected-leaves were characterized by necrosis along the veins (Fig. 6a), while infected immature cashew apples showed cracks at the epicarp limit (Fig. 6b).

|

|

|

|
The presence of pinkish-red spots, well-delimited, is characteristic of pestalotiosis on leaves (Fig. 7a). On ripe apples, long pinkish-red cracks with regular edges were observed (Fig. 7b).

|
| Table 2: | Mean incidence (%) of fungal diseases surveyed | |||
| Prefectures | Leaf anthracnose |
Fruit anthracnose |
Bud dieback | Gummosis | Black rust | Powdery mildew |
Yellow leaf spot |
Pestalotiosis | Ramularia |
| Anie | 28.75±25.88a | 0.00±0.0a | 16.25±32.81ab | 11.25±29.85a | 2.50±3.78a | 0.63±1.77a | 51.88±27.40a | 8.13±12.23 | 1.88±3.72a |
| Est-Mono | 37.50±14.64a | 24.38±36a | 58.75±25.88c | 0.00±0.0a | 27.50±15.81b | 16.88±14.87ab | 41.25±36.32a | 49.38±33.85b | 0.00±0.0a |
| Tchamba | 51.25±21.67a | 20.00±16a | 46.88±26.98bc | 0.00±0.0a | 43.13±11.93b | 20.0±12.25b | 23.13±25.62a | 87.5±21.71c | 0.00±0.0a |
| Tchaoudjo | 37.5±19.64a | 13.13±24a | 0.00±0.0a | 0.63±1.77a | 35.00±23.30b | 18.13±19.81ab | 23.75±19.59a | 99.38±1.71c | 0.00±0.0a |
| Mean | 38.75±21.44 | 14.38±23.65 | 30.47±33.56 | 2.97±15.02 | 27.03±21.28 | 13.91±15.33 | 35.0±29.24 | 61.09±41.44 | 0.47±1.95 |
| Means followed by a different letter are significantly differentat an alpha level of 0.05 according to a Tukey’stest | |||||||||
| Table 3: | Mean severity (%) of fungal diseases surveyed | |||
| Prefectures | Leaf anthracnose |
Fruit anthracnose |
Bud dieback | Gummosis | Black rust | Powdery mildew |
Yellow leaf spot |
Pestalotiosis | Ramularia |
| Anie | 1.48±0.57a | 1.00±0.0a | 1.54±1.25ab | 1.01±0.02a | 1.03±0.04a | 1.01±0.02a | 1.81±0.64a | 1.12±0.19a | 1.04±0.07a |
| Est-Mono | 1.58±0.26a | 1.53±0.80a | 2.21±0.63b | 1.00±0.0a | 1.39±0.24b | 1.30±0.29a | 1.71±0.70a | 1.63±0.42a | 1.00±0.0a |
| Tchamba | 1.93±0.54a | 1.33a±0.28a | 1.91±0.58ab | 1.00±0.0a | 1.58±0.21b | 1.33±0.21a | 1.33±0.42a | 2.58±0.79b | 1.00±0.0a |
| Tchaoudjo | 1.59±0.39a | 1.27±0.55a | 1.00±0.0a | 1.01±0.04a | 1.49±0.34b | 1.36±0.40a | 1.41±0.44a | 3.6±0.32c | 1.00±0.0a |
| Mean | 1.65±0.47 | 1.28±0.52 | 1.67±0.85 | 1.01±0.02 | 1.37±0.31 | 1.25±0.29 | 1.57±0.57 | 2.23±1.07 | 1.01±0.04 |
| Means followed by a different letter are significantly different at an alpha level of 0.05 according to a Tukey’s test | |||||||||
Ramularia was characterized by the presence of irregular pale green or yellowish spots visible on both sides of infected leaves. These spots were bounded the veins (Fig. 8a-b).
Mean incidence of fungal diseases: Regarding the mean incidence of fungal diseases (Table 2), pestalotiosis (61.09%) had the highest mean incidence, followed by leaf anthracnose (38.75%), yellow leaf spot (35%), bud dieback (30.47%), black rust (27.03%), fruit anthracnose (14.38%), powdery mildew (13.91%), gummosis (2.97%) and ramularia (0.47%).
Mean severity of fungal diseases: The data on mean severity (Table 3) showed that pestalotiosis had a slight symptom (grade 2), unlike the fungal diseases recorded, whose symptoms were minor (grade 1).
Mean severity indices for fungal diseases: The mean severity indices for pestalotiosis, bud rot and leaf anthracnose were 36.56, 20.46 and 20.16%, respectively. These were high compared to the severity indices for yellow leaf spot (14.84%), fruit anthracnose (10%), black rust (9.06%), powdery mildew (7.81%), ramularia (0.31%) and gummosis (0.15%) (Table 4).
Mean prevalence of fungal diseases: As shown in Table 5, leaf anthracnose (96.88%) was the most prevalent fungal disease in the study area, followed by pestalotiosis and yellow leaf spot (84.38%), black rust (81.25%), powdery mildew (65.63%), bud dieback (56.25%), fruit anthracnose (46.88%), gummosis (9.38%) and ramularia (6.25%).
|
| Table 4: | Mean severity index (%) of fungal diseases surveyed | |||
| Prefectures | Leaf anthracnose |
Fruit anthracnose |
Bud dieback | Gummosis | Black rust | Powdery mildew |
Yellow leaf spot |
Pestalotiosis | Ramularia |
| Anie | 14.37 | 0 | 14.37 | 0 | 0 | 0 | 20.62 | 2.5 | 1.25 |
| Est-Mono | 15.62 | 19.37 | 38.75 | 0 | 8.75 | 11.25 | 17.5 | 11.25 | 0 |
| Tchamba | 31.25 | 11.25 | 28.75 | 0 | 13.75 | 9.37 | 8.75 | 45 | 0 |
| Tchaoudjo | 19.37 | 9.37 | 0 | 0.62 | 13.75 | 10.62 | 12.5 | 87.5 | 0 |
| Mean | 20.16 | 10 | 20.46 | 0.15 | 9.06 | 7.81 | 14.84 | 36.56 | 0.31 |
| Table 5: | Mean prevalence (%) of fungal diseases surveyed | |||
| Prefectures | Leaf anthracnose |
Fruit anthracnose |
Black rust | Bud dieback | Yellow leaf spot |
Pestalotiosis | Powdery mildew |
Gummosis | Ramularia |
| Anie | 87.5 | *** | 37.5 | 37.5 | 100 | 50 | 12.5 | 25 | 5 |
| Est-Mono | 100 | 50 | 87.5 | 100 | 87.5 | 87.5 | 87.5 | *** | *** |
| Tchamba | 100 | 75 | 100 | 87.5 | 62.5 | 100 | 87.5 | *** | *** |
| Tchaoudjo | 100 | 62.5 | 100 | *** | 87.5 | 100 | 75 | 12.5 | *** |
| Mean | 96.88 | 46.88 | 81.25 | 56.25 | 84.38 | 84.38 | 65.63 | 9.375 | 6.25 |
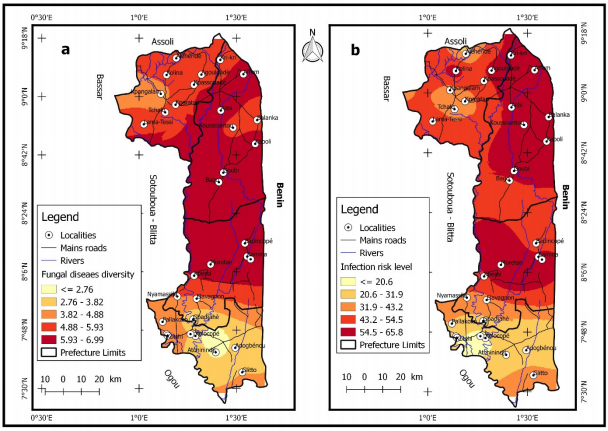
Spatial distribution of fungal diseases: Figure 9a-b illustrates the diversity of fungal diseases in our study area according to localities and risk infection levels. Yellow, yellow ochre, orange, light red and dark red colours indicate the presence of three, four, five, six and seven fungal diseases, respectively. In addition, these same colours indicate an infection risk of 12%, 13-17%, 18-23%, 24-30% and 30-42% of cashew trees, respectively.
DISCUSSION
In this study, an inventory of cashew fungal diseases was carried out and their incidence, severity and prevalence were determined in four prefectures. Eight (08) fungal diseases were inventoried and their incidence, severity and prevalence varied from one prefecture to another. Among these diseases, pestalotiosis, bud dieback, anthracnose and yellow leaf spot were the most important fungal diseases in terms of impact on the cashew cultivars exploited. Indeed, the results of the severity indices showed that for each of these four fungal diseases, at least 15% of the cashew trees inspected showed severe symptoms.
Anthracnose, yellow leaf spot, bud dieback, powdery mildew, black rust, pestalotiosis and gummosis have previously been reported in cashew orchards in Togo1,17. Their spatial distribution was uneven from one locality to another. Environmental factors or farming practices may have created favourable conditions for the establishment and spread of these fungal diseases. Similar studies conducted in Benin, Burkina Faso, Ghana, Côte d'Ivoire, Nigeria and Guinea-Bissau reported the presence of these different fungal diseases3,5,8,18. Ramularia is a cotton leaf disease caused by Ramularia areola. Its presence on some cashew leaves is explained by the susceptibility of cashew trees to attack by Ramularia areola. Indeed, it has been documented that cotton was grown in orchards where ramularia was observed or on surrounding plots. According to Pizzato et al. 19, the litter can shelter Ramularia areola from one year to the next and maybe the starting point of a primary infection at the beginning of the season. These diseases cause physio-morphological disorders in the infected parts of the cashew trees.
Several studies have shown that anthracnose, bud dieback, yellow leaf spot and black rust were the major fungal diseases of cashew trees in Togo in terms of incidence1,17. The incidence of anthracnose and bud dieback in Est-Mono prefecture was similar to those reported by Domedjui et al.1, which were 37.14 and 60%, respectively. In Tchamba prefecture, the incidences of black rust, bud dieback and yellow leaf spot were higher than those reported by Banito et al.17, which were 1.11, 15.99 and 7.83%, respectively. According to Dooh et al.20, the incidences of anthracnose, pestalotiosis, bud dieback and powdery mildew in Cameroon were 74.37, 88.62, 69.48 and 24.04%, respectively. The variation in fungal disease incidence from one prefecture to another could be explained by soil types, farming practices and biotic factors. According to Wonni et al.3 and Domedjui et al.1, the harmonious growth of plants is linked to several factors, including climatic conditions, soil types, varieties, cultural practices and the sanitary state of the crop plots.
This study's results on severity and severity indices show that pestalotiosis is the main threat to the imminent decline of cashew nut production in the prefectures surveyed. The pestalotiosis severity index mean was higher than 15.52% for red-fruited (AB05-08) and yellow-fruited (AB 29) varieties20. This difference could also be related to the susceptibility of the cashew cultivars used. According to Palei et al.21 wild cashew genotypes are more susceptible to diseases than improved cultivars. Climatic conditions inside cashew orchards could also influence the pathogenicity of Pestalotia heterocornis strains, the causative agent of pestalotiosis. Therefore, cashew cultivars should be selected on the basis of their productive capacity and their resistance to diseases. The anthracnose severity index means was in agreement with Dooh et al.20 who reported that it was 18.55%. In addition, the same authors reported that the average severity index for bud dieback and powdery mildew were 12.28 and 4.54%, respectively. This shows that the latter is lower than this study’s results. This difference in severity indices could be related to the vigour of the cashew trees inspected. Indeed; the age of the cashew trees inspected in this study ranged from 5 to 48 years, while those inspected in Cameroon were 10 years old.
Leaf anthracnose, yellow leaf spot, pestalotiosis, black rust, powdery mildew and bud dieback all had a prevalence of over 50%. This indicates a wide spatial distribution of these diseases. The prevalence of bud dieback in the Tchamba prefecture was similar to that reported by Banito et al.17, which was 100%, making it the most widespread fungal disease of cashew trees. Similarly, the mean prevalences of anthracnose and bud dieback in the Est-Mono prefecture were similar to those obtained previously1. They were 71.43 and 100%, respectively. The increasing dynamic of fungal disease prevalence’s over time reflects their continuous spread in the orchards. This could be explained by a number of factors, in particular the lack of phytosanitary treatments. In fact, for the vast majority of cashew producers interviewed, physical control is the main disease control measure. This observation is in contrast to that made in the Tanga region of Tanzania, where the majority (45%) of cashew producers control pests and diseases mainly with pesticides22.
The cashew cultivars exploited are susceptible to attack by pathogenic fungi. This study’s results provide a scientific database that could guide political authorities or NGOs in any cashew disease management program. For new orchards, the improved cultivars are recommended based on epidemiological data. Further studies on cashew pathogens should increase the number of individuals inspected and orchards surveyed to minimize standard errors.
CONCLUSION
This study involved an inventory of cashew tree fungal diseases in the prefectures of Anié, Est-Mono, Tchamba and Tchaoudjo in Togo. Eight fungal diseases were reported such as anthracnose, black rust, bud dieback, yellow leaf spot, pestalotiosis, powdery mildew, gummosis and ramularia. These fungal diseases weren’t evenly distributed over the study area. Pestalotiosis and ramularia were newly reported on cashew trees. The mean incidence, severity index, severity and prevalence of the inventoried fungal diseases varied from one prefecture to another. In terms of incidence and severity, pestalotiosis was the main fungal disease of cashew trees, followed by leaf anthracnose, bud dieback, yellow leaf spot and black rust. The use of phytosanitary products and resistant cultivars will contribute to the control of the cashew fungal pathogens to reduce the loss of productivity of the cashew nut in Togo.
SIGNIFICANCE STATEMENT
This study’s purpose was to draw up an inventory of cashew fungal diseases and determine their epidemiological parameters (Incidence, severity and prevalence) in Anié, Est-Mono, Tchamba and Tchaoudjo prefectures in Togo. Eight fungal diseases were inventoried, including anthracnose, black rust, bud dieback, yellow leaf spot, pestalotiosis, powdery mildew, gummosis and ramularia. These fungal diseases weren’t evenly distributed over the study area. Pestalotiosis and ramularia were newly reported on cashew trees. The mean incidence, severity, severity index and prevalence of the inventoried fungal diseases varied from one prefecture to another. The characterization of the fungal pathogens associated with these eight fungal diseases is this study’s future perspective.
ACKNOWLEDGMENT
Our thanks to Tchaou B.P.I. BODJONA, N'pagyendou LARE and Nathalie JOHNSON for their respective donations.
REFERENCES
- Domedjui, K.L., K.E. Kpemoua, E. Tedihou, K.D. Etse and E. Palanga et al., 2023. Inventory of cashew fungal diseases in Est-Mono prefecture Togo. J. Recherche Scientifique l’Université Lomé, 25: 1-11.
- Muntala, A., S.K. Gyasi, P.M. Norshie, S. Larbi-Koranteng, F.K. Ackah, D.A. Ntiamoah and M.A. Mohamed, 2021. Diseases and insect pests associated with cashew (Anacardium occidentale L.) orchards in Ghana. Eur. J. Agric. Food Sci., 3: 23-32.
- Wonni, I., D. Sereme, I. Ouedraogo, A.I. Kassankagno, I. Dao, L. Ouedraogo and S. Nacro, 2017. Diseases of cashew nut plants (Anacardium occidentale L.) in Burkina Faso. Adv. Plants Agric. Res., 6: 78-83.
- Khatoon, A., A. Mohapatra and K.B. Satapathy, 2017. Major diseases of cashew (Anacardium occidentale L.) caused by fungi and their control in Odisha, India. Int. J. Biosci., 11: 68-74.
- Monteiro, F., M.M. Romeiras, J. Barnabé, S. Catarino, D. Batista and M. Sebastiana, 2022. Disease-causing agents in cashew: A review in a tropical cash crop. Agronomy, 12.
- Freire, F.C.O., J.E. Cardoso, A.A. dos Santos and F.M.P. Viana, 2002. Diseases of cashew nut plants (Anacardium occidentale L.) in Brazil. Crop Prot., 21: 489-494.
- Adejumo, T.O., 2005. Crop protection strategies for major diseases of cocoa, coffee and cashew in Nigeria. Afr. J. Biotechnol., 4: 143-150.
- Houndahouan, D.E.T., A. Zannou, R. Sikirou, A. Adomou, V. Zinsou, S. Boukari and K. N’djolossè, 2018. Economic losses due to cashew anthracnose in Benin. Eur. Sci. J., 14: 127-138.
- Dominic, M. and Marthamakobe, 2016. Biological control of cashew powdery mildew using Ampelomyces quisqualis Ces. J. Biol. Control, 30: 226-235.
- Sijaona, M.E.R., A. Clewer, A. Maddison and J.W. Mansfield, 2001. Comparative analysis of powdery mildew development on leaves, seedlings and flower panicles of different genotypes of cashew. Plant Pathol., 50: 234-243.
- Menge, D. and S. Shomari, 2016. Studies on leaf and nut blight of cashew (Anacardium occidentale) caused by Cryptosporiopsis sp. in Tanzania. Plant Pathol. Quarantine, 6: 201-205.
- Tibuhwa, D.D. and S. Shomari, 2016. Fusarium wilt disease: An emerging threat to cashew nut crop production in Tanzania. Asian J. Plant Pathol., 10: 36-48.
- Uaciquete, A., L. Korsten and J.E. van der Waals, 2013. A search for anthracnose resistant cashew cultivars in Mozambique. Crop Prot., 50: 6-11.
- Cardoso, J.E., J.R. Paiva, J.J.V. Cavalcanti, A.A. dos Santos and J.C. Vidal, 2006. Evaluation of resistance in dwarf cashew to gummosis in North-Eastern Brazil. Crop Prot., 25: 855-859.
- Moreira, R.C., J.S. Lima, L.G.C. Silva and J.E. Cardoso, 2013. Resistance to gummosis in wild cashew genotypes in Northern Brazil. Crop Prot., 52: 10-13.
- Francis, Y.K., A.K. Patrice, S. Koutoua and D.A. Hortense, 2017. Distribution and prevalence of yam anthracnose in four growing zones of Côte d’Ivoire. Int. J. Innovation Appl. Stud., 19: 463-474.
- Banito, A., E.K. Kpemoua, R.K. Dayiwo, E. Tedihou and R. Sikirou, 2021. Cashew (Anacardium occidentale L.) diseases’ inventory in the prefecture of Tchamba in Togo. Int. J. Biol. Chem. Sci., 15: 2514-2525.
- Adeniyi, D.O. and E.U. Asogwa, 2023. Dynamics of Diseases and Insect Pests of Cashew Tree. In: Forest Microbiology: Tree Diseases and Pests, Asiegbu, F.O. and A. Kovalchuk (Eds.), Academic Press, Cambridge, Massachusetts, ISBN: 9780443186943, pp: 265-284.
- Pizzato, J.A., D.V. Araújo, M.E. Serafim, K.L. Araújo and R. Dallacort et al., 2013. Epidemiologic study of Ramularia areola under different soil covers and spacings, for cotton crops. Am. J. Plant Sci., 4: 2049-2059.
- Dooh, J.P.N., C.B.D. Asta, B. Djile, D.B.T. Tchoupou and A. Heu et al., 2021. Major fungi diseases of cashew trees (Anacardium occidental L.) in Cameroon. J. Agric. Sci., 13: 124-134.
- Palei, S., R. Dasmohapatra, S. Samal and G.R. Rout, 2019. Cashew Nut (Anacardium occidentale L.) Breeding Strategies. In: Advances in Plant Breeding Strategies: Nut and Beverage Crops, Al-Khayri, J.M., S.M. Jain and D.V. Johnson (Eds.), Springer, Cham, Switzerland, ISBN: 978-3-030-23111-8, pp: 77-104.
- Nene, W., G.M. Rwegasira and M. Mwatawala, 2017. Optimizing a method for locating queen nests of the weaver ant Oecophylla longinoda Latreille (Hymenoptera: Formicidae) in cashew, Anacardium occidentale L. plantations in Tanzania. Crop Prot., 102: 81-87
How to Cite this paper?
APA-7 Style
Domedjui,
K.L., Gbogbo,
K.A., Kpemoua,
K.E., Folega,
F., Kanda,
M., Akpavi,
S., Dourma,
M., Batawila,
K., Akpagana,
K. (2024). Epidemiology of the Major Fungal Diseases of Cashew Tree (Anacardium occidentale L.) in Togo. Asian Journal of Plant Pathology, 18(1), 49-58. https://doi.org/10.3923/ajpp.2024.49.58
ACS Style
Domedjui,
K.L.; Gbogbo,
K.A.; Kpemoua,
K.E.; Folega,
F.; Kanda,
M.; Akpavi,
S.; Dourma,
M.; Batawila,
K.; Akpagana,
K. Epidemiology of the Major Fungal Diseases of Cashew Tree (Anacardium occidentale L.) in Togo. Asian J. Plant Pathol. 2024, 18, 49-58. https://doi.org/10.3923/ajpp.2024.49.58
AMA Style
Domedjui
KL, Gbogbo
KA, Kpemoua
KE, Folega
F, Kanda
M, Akpavi
S, Dourma
M, Batawila
K, Akpagana
K. Epidemiology of the Major Fungal Diseases of Cashew Tree (Anacardium occidentale L.) in Togo. Asian Journal of Plant Pathology. 2024; 18(1): 49-58. https://doi.org/10.3923/ajpp.2024.49.58
Chicago/Turabian Style
Domedjui, Komi, Lucien, Koffi Apeti Gbogbo, Kossi Essotina Kpemoua, Fousséni Folega, Madouma Kanda, Sêmihinva Akpavi, Marra Dourma, Komlan Batawila, and Koffi Akpagana.
2024. "Epidemiology of the Major Fungal Diseases of Cashew Tree (Anacardium occidentale L.) in Togo" Asian Journal of Plant Pathology 18, no. 1: 49-58. https://doi.org/10.3923/ajpp.2024.49.58

This work is licensed under a Creative Commons Attribution 4.0 International License.


